
Jiangsu East-Mab Biomedical Technology Co., Ltd, established in 2016, is a leading biotechnology company dedicated to providing high-quality recombinant protein raw materials worldwide.
Backed by renowned investment institutions and numerous industry partners, the company has gradually transitioned from antibody drug R&D to the R&D, production and sales of recombinant protein raw materials. EastMabBio mainly provides high-quality cell culture proteins, IVD diagnostic proteins, pharmaceutical / diagnostic enzyme series, as well as related technical services. Over the past few years, the company has invested more than $30 million to build a recombinant protein R&D, validation and manufacturing platform with large-scale supply capacity and in compliance with international regulatory standards. This platform supports the development of global IVD, cell culture media, cell therapy, organoids, cultivated meat and other fields.
- 2016Founded in
- 10000㎡R&D and production space
- 4000㎡GMP-standard workshop
- 1000LMaximum fermentation tank
- 150+Biopharmaceutical background team
- 60+Industrial grade shaker

 Jiangsu EastMabBio
Jiangsu EastMabBio
 Suzhou EastMabBio
Suzhou EastMabBio
GMP Compliance Statement – SGS
Quality Management System Certificate
Certificated with ISO13485, ISO9001, ISO14001 and ISO45001




DMF Filing
DMF(FDA) filings for 21 cell culture proteins
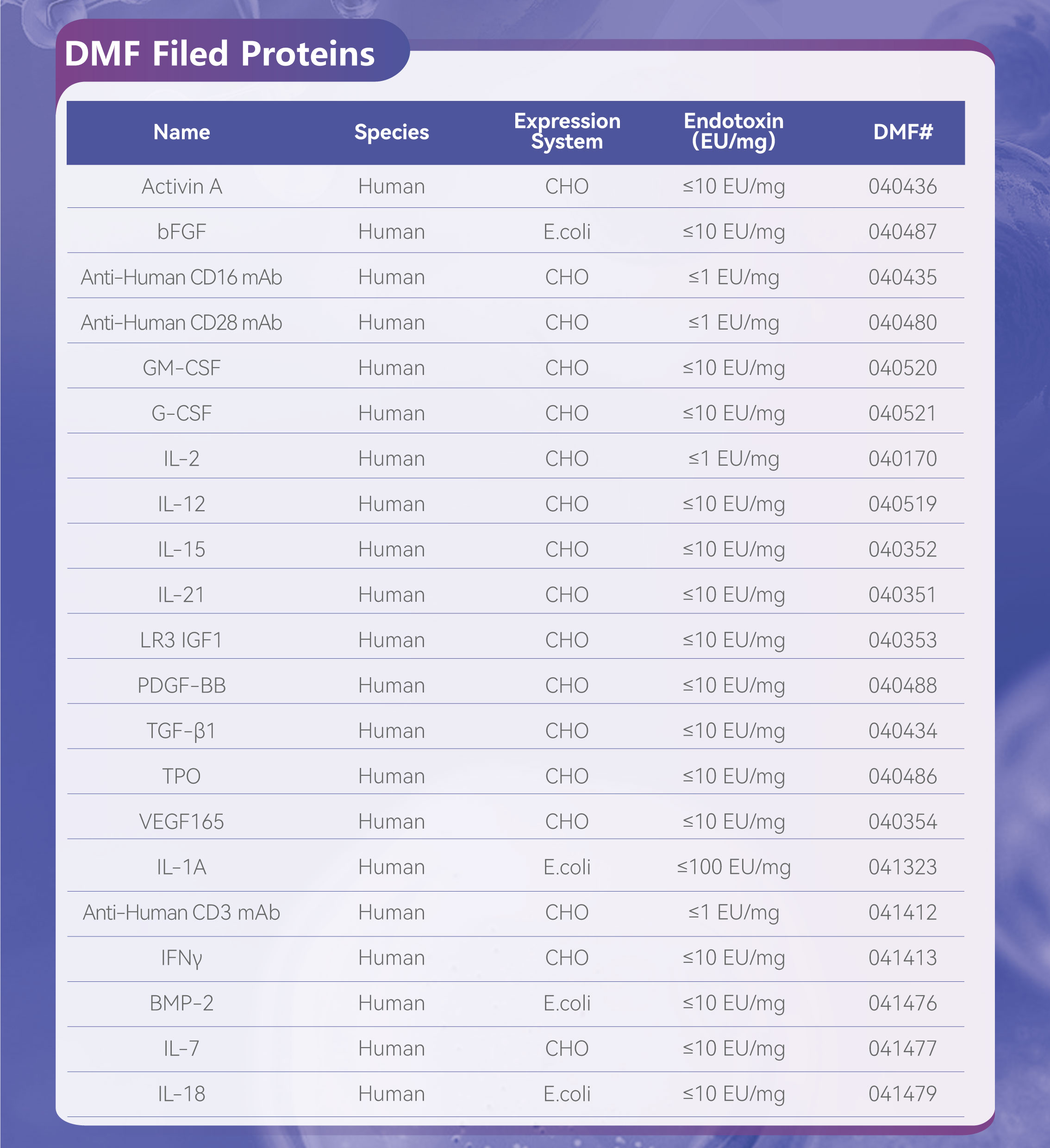
The data source is up to date as of April 21, 2025.
GMP Workshop
Operating 4000m² GMP-standard workshop (additional details to follow)




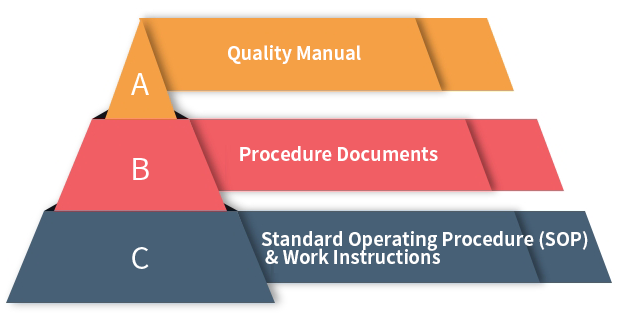
Quality Management Documents
Strict quality and production process control
Validated analytical methodologies
Cell culture protein raw materials analytical methods:
• Bioactivity: developed nearly 50 cell banks dedicated to testing the bioactivity of various cell culture proteins, with further expansion underway
• Stability Testing: evaluate lot-to-lot consistency and product performance over time
• SDS-PAGE
• HPLC
• UV Spectrophotometry
• Endotoxin Quantification
• Microbial Contamination Testing
• Other Quality Indicators: includes assessment of appearance, hydration, turbidity, content.
IVD diagnostic protein raw materials analytical methods:
• UV Spectrophotometry
• SDS-PAGE
• HPLC
• Colloidal Gold Assay
• LC-MS
• SPR
• Sterility Testing
• Dynamic Chromogenic Assay
• Other analyses are conducted as needed to ensure comprehensive quality control.
Medical & Diagnostic enzyme raw materials analytical methods:
• SDS-PAGE
• UV Spectrophotometry
• HPLC
• Agarose Electrophoresis
• qPCR
• Real-Time
• Other specialized tests are conducted as required for comprehensive quality assurance.
 MissionImprove health
MissionImprove health PositionHigh-quality Recombinant Protein Manufacturer
PositionHigh-quality Recombinant Protein Manufacturer DedicationTechnology-Driven, Open Communication, Conscientious and Responsible
DedicationTechnology-Driven, Open Communication, Conscientious and Responsible



































































